Precise temperature measurements are of critical importance in many industrial sectors, particularly in the pharmaceutical industry, cryogenics, and scientific research.
The calibration of thermometers at extremely low temperatures, ranging from -80 °C to -180 °C, represents a significant technical challenge. This article describes the technical possibilities for calibrating thermometers at these extremely low temperatures.
Content
Calibration procedures at low temperatures
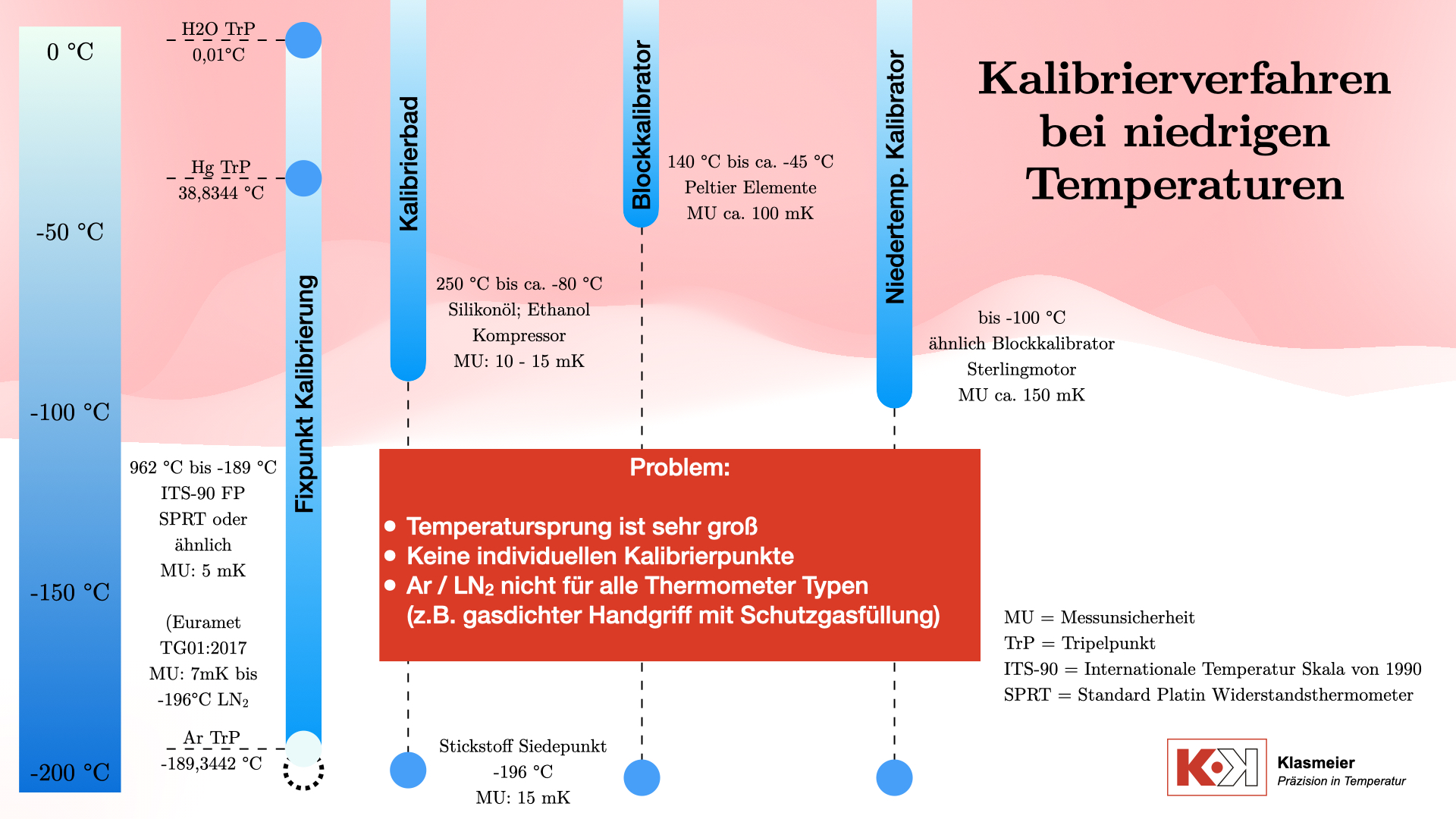
Calibration baths
The classic procedure for calibrating thermometers at low temperatures uses calibration baths. These typically consist of two working chambers:
- Calibration medium chamber: In this chamber, the calibration medium is tempered and circulated.
- Calibration chamber: The thermometer is calibrated in this chamber.
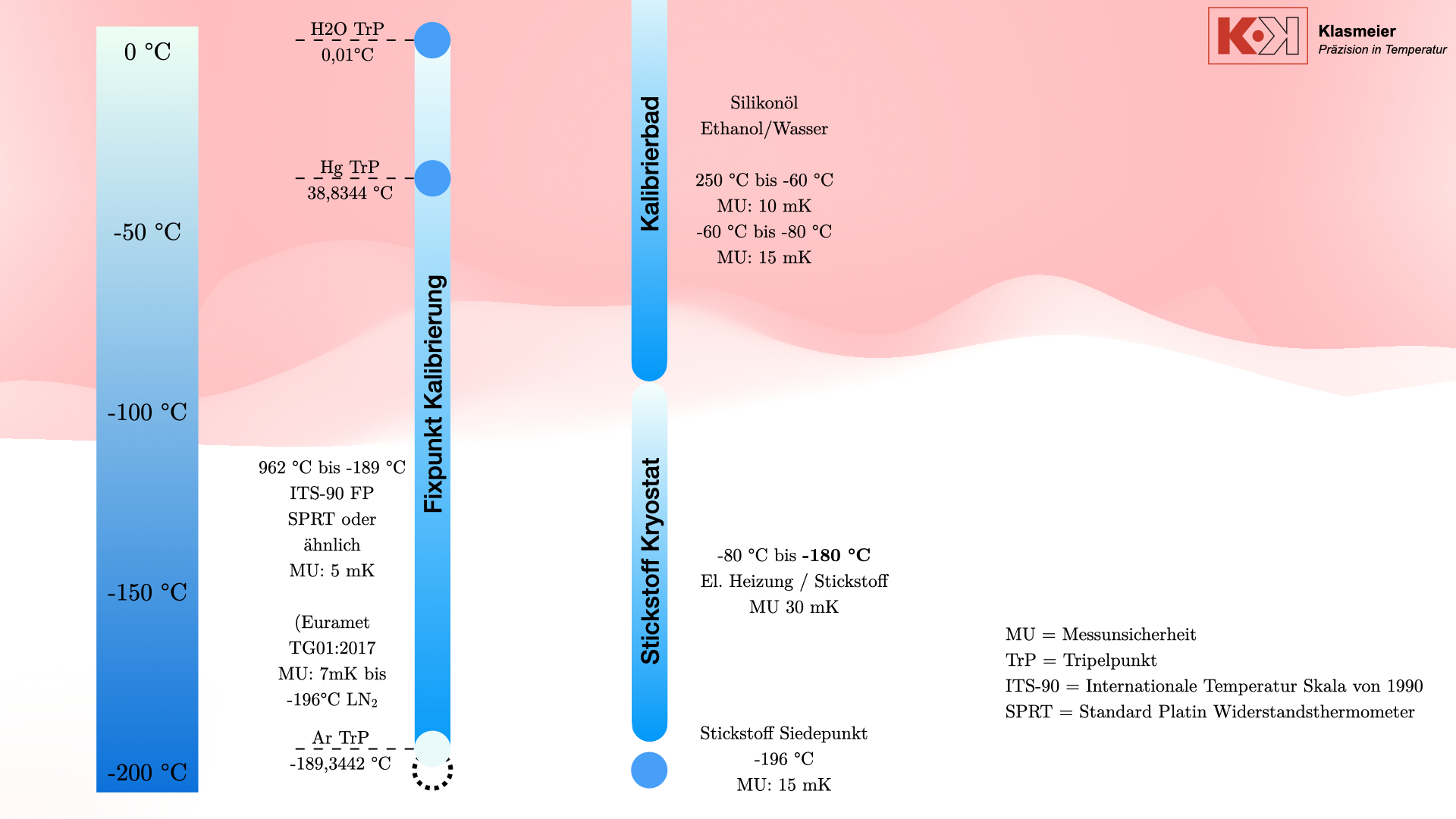
The most common calibration medium is silicone oil, which is used for temperatures up to 250 °C. For lower temperatures, down to approximately -80 °C, ethanol is used. Some calibration baths can reach even lower temperatures, but the effort is often disproportionate to the benefit. Calibration baths are cooled by means of a compressor. This compressor always runs at 100% power, and an electric heater tempers against this full cooling capacity. The calibration temperature is therefore not reached by cooling, but by heating against the cooling. This has the advantage that a significantly higher control accuracy or stability can be achieved. The disadvantage of this technology is the great effort required to reach a stable temperature.
The achievable measurement uncertainty of these calibration baths is typically 10 mK to 15 mK.
Temperature block calibrators
Temperature block calibrators are widely used in industry due to their ease of use. They work with Peltier elements and can reach temperatures up to about -70 °C below ambient temperature. As a result, the temperature range in the negative area is limited to about -45 °C when used in laboratory environments. Their measurement uncertainty is approximately 50 mK to 100 mK, which is sufficient for many industrial applications. For use in a calibration laboratory, this measurement uncertainty is not precise enough.
Low-temperature calibrators
Low-temperature calibrators are similar in design to standard temperature block calibrators. However, they do not work with Peltier elements but use Stirling engines to cool the calibration volume. They can reach temperatures down to -100 °C. These devices have the advantage that no liquids are used as a calibration medium. However, they are relatively expensive and have a high measurement uncertainty of about 150 mK.
Calibration at the secondary temperature fixed point of nitrogen
In addition to classic calibration procedures, there is the possibility of calibrating thermometers at the secondary temperature fixed point of nitrogen. The boiling point of nitrogen is -196 °C and represents a well-defined secondary fixed point that can be used technically for calibration. However, this procedure also has some disadvantages.
Although calibration at the nitrogen boiling point is technically feasible and can be implemented very cost-effectively, this point is not defined in the International Temperature Scale of 1990 (ITS-90). This means that the calibration does not achieve the same accuracy and international recognition as temperature fixed-point calibration according to ITS-90.
Another problem is the limited number of calibration points. When using the nitrogen boiling point, too few calibration points are available. The temperature gap from the approx. -80 °C or -100 °C in, for example, a calibration bath to the nitrogen boiling point at -196 °C is too large to calculate a precise thermometer characteristic curve. Accurate and continuous calibration over the entire temperature range is therefore not possible.
Calibration at the nitrogen boiling point is therefore not recommended, even though it is technically feasible.
Temperature fixed-point calibration according to ITS-90
The most precise method for calibrating thermometers is fixed-point calibration according to the International Temperature Scale of 1990 (ITS-90). This method includes the following temperature fixed points in the negative temperature range:
- Triple point of water: 0.01 °C
- Triple point of mercury: -38.8344 °C
- Triple point of argon: -189.3442 °C
Additionally, the nitrogen boiling point at -196 °C can be used as a secondary temperature fixed point. With the EURAMET TG 01:2017 guideline, the ITS-90 thermometer characteristic curve (deviation function) can then be extrapolated down to -196 °C.
A major problem with calibration at extremely low temperatures at temperature fixed points is the handling of the thermometers. Special gas-tight handles and inert gas fillings are necessary to ensure precise measurements.
This method covers the entire temperature range, but has the disadvantage that not all thermometers are suitable for fixed-point calibration. This leads to a gap in calibration capability for certain temperature ranges and thermometer types. The thermometers must be capable of being calibrated at temperature fixed points in terms of their design, i.e., they must have a minimum length and a suitable diameter on the protection tube.
Furthermore, there is the major restriction that the thermometers must be exposed to the extremely low temperature of approx. -189 °C at the triple point of argon. For this to be possible, certain design requirements for the thermometer are necessary. For example, there must be no air in the measuring channel of the thermometer, as the phase transition of oxygen would make calibration impossible. This fact restricts many thermometers. A thermometer that, for example, may only be used down to -150 °C is therefore not capable of being calibrated at temperature fixed points in the negative range.
Challenge: The phase transition of air
A central problem in calibration at extremely low temperatures is the phase transition of air. At temperatures below -180 °C, the components of air (mainly nitrogen and oxygen) begin to condense and eventually liquefy. This leads to significant measurement problems as the gas mixture exists in different phases.
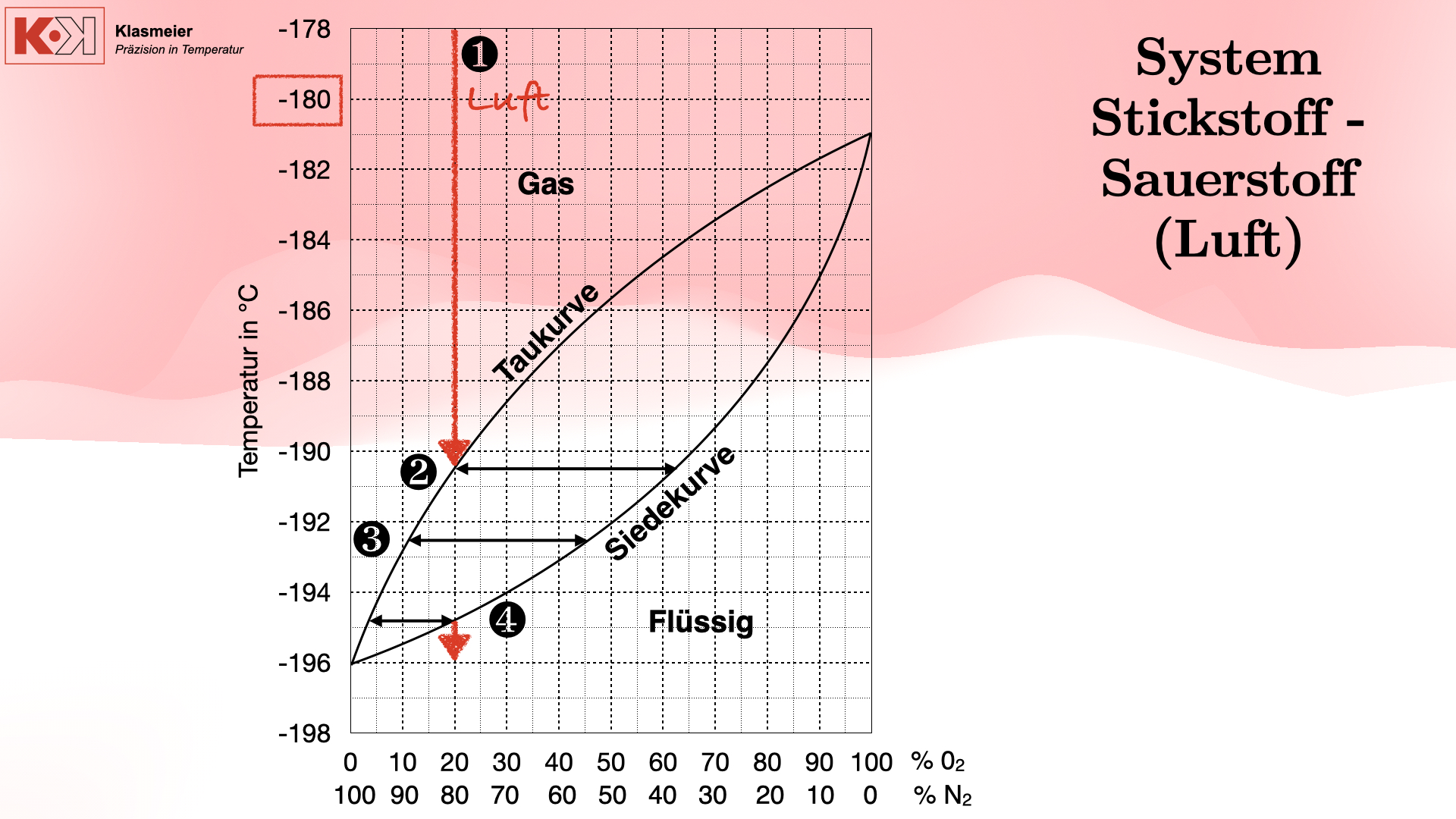
Phase transition of air:
- Dew point: When air cools, droplets of liquid oxygen and nitrogen first form, whereby the dew point is not uniform because it is a gas mixture.
- Boiling point: Further cooling leads to the complete liquefaction of the air components, but with different compositions.
- Condensation: Finally, the air condenses completely into a liquid mixture of 80% nitrogen and 20% oxygen.
This phase transition can cause thermometers passed through this range to provide unreliable measurements as they are subject to various phase transitions.
Solution: Inert gas and gas-tight handles
The phase transition of air must be prevented in the thermometer, as an air-filled thermometer does not function at extremely low temperatures. One way to solve this problem is to fill the thermometers with inert gas:
- Inert gas filling: We use an inert gas (e.g., helium or argon) in the thermometer to prevent the phase transition. Helium and argon have very low boiling points and remain gaseous at the calibration temperatures, ensuring the stability of the measurements.
- Gas-tight handles: Our thermometers are equipped with gas-tight handles. These handles are designed to be completely sealed and keep the inert gas inside. This prevents ambient moisture or air from entering the thermometer and triggering the phase transition.
The nitrogen cryostat
To bridge the gap between classic thermometer calibration procedures and temperature fixed-point calibration, nitrogen cryostats can be used.
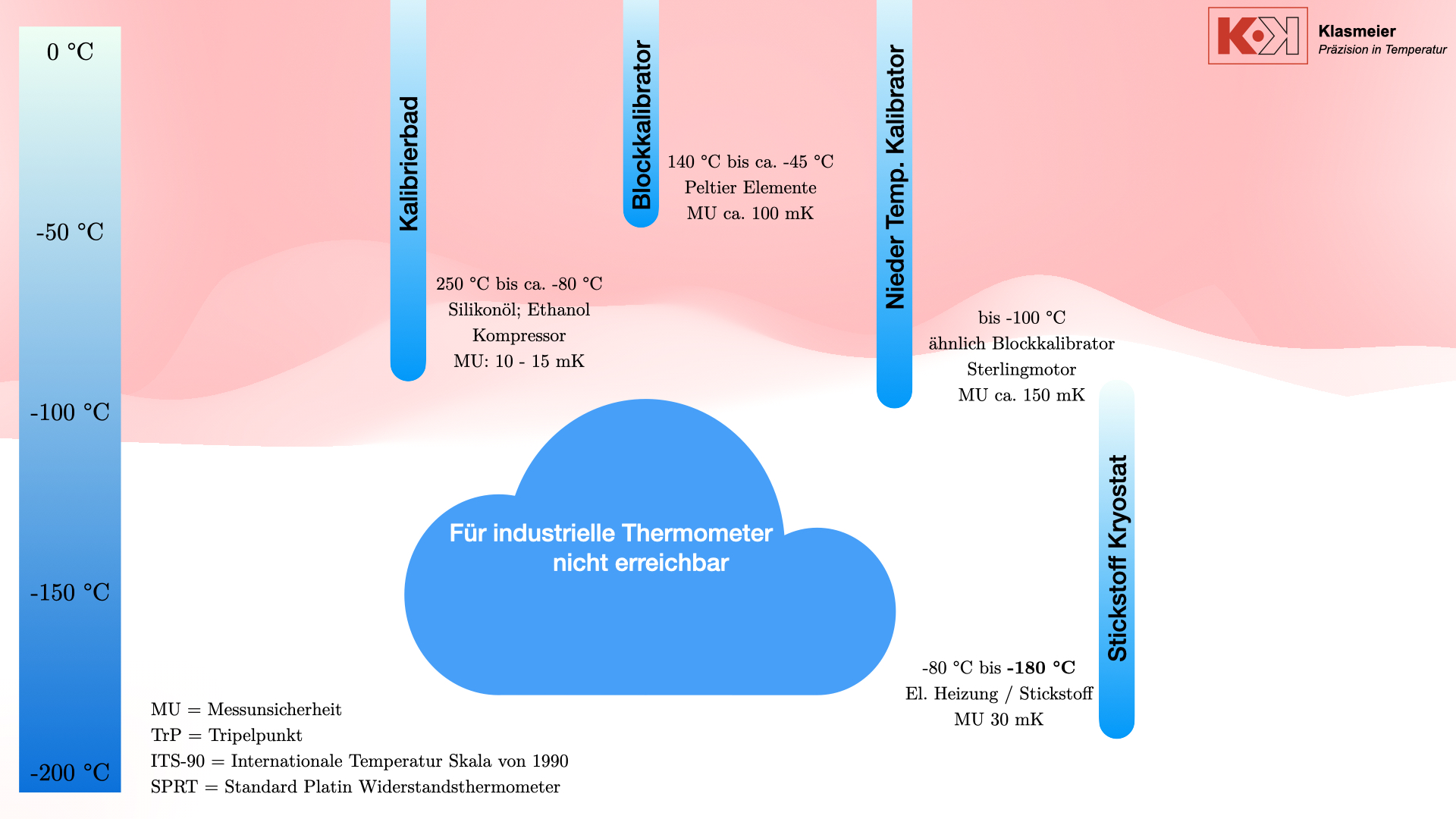
This system uses liquid nitrogen and an electric heater to achieve stable calibration temperatures, offering a flexible and precise solution for calibration at extremely low temperatures.

Design and principle of operation
The nitrogen cryostat consists of the following components:
- Stainless steel vessel: Gas-tight welded and placed on a base so that it can be surrounded by liquid nitrogen.
- Copper calibration insert: Contains two stainless steel thermometer sleeves that are gas-tight welded.
- Electric heater: To heat the nitrogen in a targeted manner and achieve stable calibration temperatures.
- Vacuum or inert gas system: To prevent the phase transition of air and ensure a stable calibration temperature.

By creating a vacuum or filling with an inert gas such as argon or helium, the phase transition of air is prevented from affecting the calibration. The cryostat can reach temperatures from -80 °C to -180 °C with a measurement uncertainty of 30 mK.
Evacuation of the cryostat:
The phase transition of air must be prevented not only in the thermometers but also in the cryostats. Therefore, the cryostat must also be filled with inert gas.
- Evacuation: The stainless steel vessel of the cryostat is first evacuated to remove the air.
- Filling with inert gas: The vessel is then filled with inert gas (e.g., helium).
- Sealing: The thermometers are inserted into the gas-tight sleeves and sealed with special Teflon seals to prevent the ingress of moisture.
- Calibration: The thermometer is brought to the desired calibration temperature in the nitrogen cryostat and calibrated.
Practical application and integration
Nitrogen cryostats can be integrated into existing calibration facilities. In combination with cryostats and calibration baths, a thermometer that does not meet the requirements for calibration at temperature fixed points can be completely calibrated in the negative temperature range.
The nitrogen consumption for a complete calibration is approximately 60 liters, which is sufficient for one week of operation. The initial cooling time of the cryostat is about six hours, and temperature changes take up to four hours depending on the temperature difference.
Example of a calibration
A typical calibration process involves several steps:
- Initial cooling: The cryostat is filled with liquid nitrogen and cooled to -196 °C.
- Temperature stabilization: The desired calibration temperature is reached by targeted heating against the boiling point of nitrogen.
- Calibration: The thermometer to be calibrated is inserted into the thermometer sleeves, and the temperature is measured and compared. Temperature differences or pairs of values are determined, and the thermometer under test is calibrated.

